Petromyzonti (lampreys) >
Petromyzontiformes (Lampreys) >
Petromyzontidae (Northern lampreys) > Lampetrinae
Etymology: Lethenteron: Etymology not explained, perhaps lethalis (L.), lethal, or lethe (Gr.), forgetting or forgetfulness; enteron (Gr.) intestine, presumably referring to “degenerate and non-functional” intestine of adult L. appendix (See ETYFish); kessleri: Patronym not identified but probably in honor of German-Russian zoologist Karl Fedorovich Kessler (1815-1881), who described Caspiomyzon wagneri in 1870 (See ETYFish).
Eponymy: Karl Fedorovich (Theodorovich) Kessler (1815–1881) was a Russian-German zoologist and collector who was one of the founders of the St Petersburg Society of Naturalists (1868), and its President (1868–1879). [...] (Ref. 128868), visit book page.
Environment: milieu / climate zone / depth range / distribution range
Ecology
Freshwater; demersal; non-migratory. Temperate; 5°C - 25°C (Ref. 12468); 78°N - 53°N, 55°E - 169°W
Asia: Throughout Siberia to the Anadyr and Sakhalin (Ref. 26334). Specimens identified as Lethenteron kessleri from the Arctic basin refer to Lethenteron reissneri (Ref. 59043).
Length at first maturity / Size / Weight / Age
Maturity: Lm 12.4, range 11 - 13.27 cm
Max length : 26.0 cm TL male/unsexed; (Ref. 12323); max. reported age: 7 years (Ref. 12323)
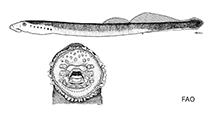
Adults: 11.2-23.0 cm TL. Body proportions, as percentage of TL (based on 300 specimens measuring 15.3-23.0 cm TL; the values given below represent ranges of means based on samples of 50 specimens): prebranchial length, 11.2-12.0; branchial length, 9.5-9.6 (8.9-11.9 absolute range, Iwata et al. (1985) based on 97 specimens 11.2-18.4 cm TL); trunk length, 48.8 (derived by deduction); tail length, 29.1-30.9; eye length, 2.1-2.2; disc length, 5.6-6.0. The intestinal diameter is less than 1.0 mm. Urogenital papilla length, as a percentage of branchial length, in three spawning males measuring 14.7-16.9 cm TL, 5.9-10.0 [5.8-34.7, Iwata et al. (1985) based on 23 spawning males 12.1-16.0 cm TL]. Succeeding counts/descriptions in parentheses are of resident L. camtschaticum form sensu Kucheryavyi et al. (2007): Trunk myomeres, 65-73 (57-78). Dentition: supraoral lamina, 2 unicuspid teeth, rarely 3; infraoral lamina, 5-10 teeth, usually 6-8, the lateralmost unicuspid or bicuspid (one or both rarely unicuspid) and occasionally some of the internal ones also bicuspid, the rest unicuspid; 3 endolaterals on each side; endolateral formula, typically 2-2-2, variant formulae, 2-2-1 and 2-1-2 (also 2-2-2-2); 2 rows of anterials; first row of anterials, 3-5 unicuspid teeth; total number of anterials, 15-28 (8-30) unicuspid teeth; exolaterals usually absent, but if present, only 1 unicuspid tooth per lateral field; single row of posterials consisting of 16-25 (as low as 12) unicuspid teeth, sometimes completely absent; transverse lingual lamina, numerous unicuspid teeth, the median one enlarged; longitudinal lingual laminae parentheses-shaped, each with numerous unicuspid teeth. Velar tentacles, 7. Body coloration of live specimen, brownish on dorsal and lateral aspects and whitish on the ventral aspect. Second dorsal fin with a dark blotch near the apex; however, specimens from Japan do not have a dark blotch. Extent of caudal fin pigmentation, rarely absent or trace, usually 75% or more. Caudal fin shape, spade-like.
Body shape (shape guide): eel-like.
In Europe the upper courses of rivers, in Siberia also in lakes and lowland rivers (Ref. 12323). Freshwater, in rivers, brooks, and lakes. Ammocoetes live in oozy substrate. Metamorphosing ammocoetes are mostly found in areas with abundant aquatic vegetation at the mouth of small streams (Ref. 89241). Ammocoetes feed on detritus, green algae, diatoms, and zooplankton (Ref. 12323. 89241). Larval life lasts 6+ yrs. Onset of metamorphosis is at the end of July beginning of August and is complete at the end of October to the middle of November. The population in the upper Irtysh River Basin is apparently unique among lampreys in that it continues to feed during metamorphosis gradually switching from a detritus and unicellular algae diet to an exclusively unicellular algae diet. This would explain the fact that the maximum total length of the metamorphosing ammocoete exceeds the maximum total length of the ammocoete; mean of 233 mm in the former versus 218 in the latter. Unicellular algae consumed include Cyanophyceae (Oscillatoria), Chlorophyceae (Spirogyra, Chlorella), and Diatomeae (Fragilaria, Pinnularia). This phenomenon is particularly interesting as it implies that the metamorphosing ammocoetes must switch from a filter-feeding mode to a grazing mode of feeding, since, during the process of metamorphosis, the oral cirrhi are lost. The grazing may be achieved possibly with the help of the lingual laminae and the action of the piston cartilage (Ref. 89241). Adults are nonparasitic (Ref. 89241, 12323). Spawning period in Japan in early May. Fecundity, 1,387-2,125 eggs/female in Japan, with long diameter of eggs varying from 1.12 to 1.22 mm and the short diameter from 0.91 to 1.11 mm. Kucheryavyi et al. (2007) reported a fecundity of 468-3,441 eggs/female in non-feeding adult stage resident L. camtschaticum (= L. kessleri?) on Kamchatka (Ref. 89241). Ammocoetes used as lures for bait casting (Ref. 12323).
Holcík, J., 1986. Lethenteron kessleri (Anikin, 1905). p. 220-236. In J. Holcík (ed.) The Freshwater fishes of Europe. Vol.1, Part I, Petromyzontiformes. (Ref. 12323)
IUCN Red List Status (Ref. 130435: Version 2025-2)
Threat to humans
Harmless
Human uses
Fisheries: of no interest; bait: usually
Tools
Special reports
Download XML
Internet sources
Estimates based on models
Phylogenetic diversity index (Ref.
82804): PD
50 = 0.5020 [Uniqueness, from 0.5 = low to 2.0 = high].
Bayesian length-weight: a=0.00138 (0.00062 - 0.00310), b=2.97 (2.78 - 3.16), in cm total length, based on LWR estimates for this (Sub)family-body shape (Ref.
93245).
Trophic level (Ref.
69278): 2.2 ±0.14 se; based on food items.
Resilience (Ref.
120179): Low, minimum population doubling time 4.5 - 14 years (tmax=tm=7).
Fishing Vulnerability (Ref.
59153): Low vulnerability (16 of 100).
🛈